What is Covalent Bonding?
Covalent bonding is a type of bonding formed only between non-metal elements with some exceptions. This bonding is formed by sharing a pair of an electron to achieve a full outmost shell. But the question is why does sharing a pair of electrons make the atoms stick together is a covalent bond?
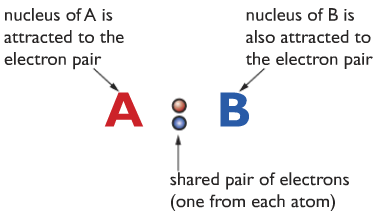
Suppose two atoms A and B are going to form a covalent bond. They approach each other. When they get closer together, their outer shell electron feels attraction towards the nucleus of the other atom. The electron density shifts so the two electrons are between the two nuclei
The nuclei of the two atoms feel an attraction towards the shared electron pair, which causes them to stick together in a covalent bond.
What is bond length?
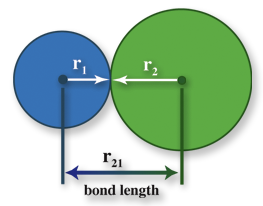
But also, the two nuclei feels repulsion towards each other also. For this, they need to stay a certain distance apart from each other, so there is a balance between the attractive and repulsive forces. The separation distance between the two nuclei is known as bond length. For example, in a hydrogen molecule, the bond length is 0.074 nm.
What is bond enthalpy?
When the covalent bond is formed, energy is released. This is known as bond enthalpy. Same amount of energy is required to break the bond. The bond enthalpy of H2 molecule is 436 Kmol-1
Different representations of covalent bonds
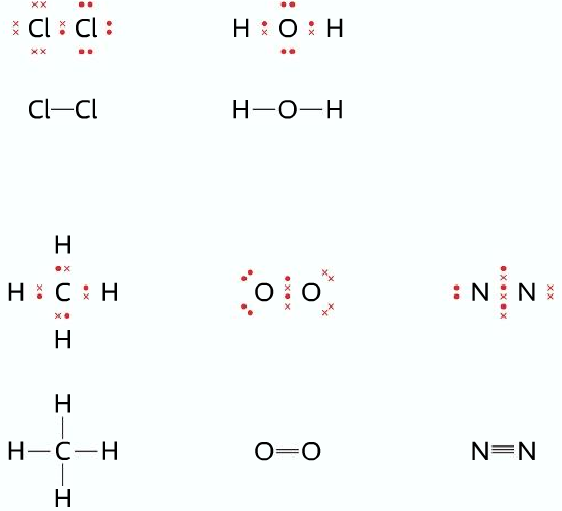
When one electron pair is shared, one covalent bond is formed. The covalent bond can be represented by a dot-cross diagram or by structural formula, where a single covalent bond is represented by a solid line. Please check out the diagram below:
What is Dative Covalent Bonding?
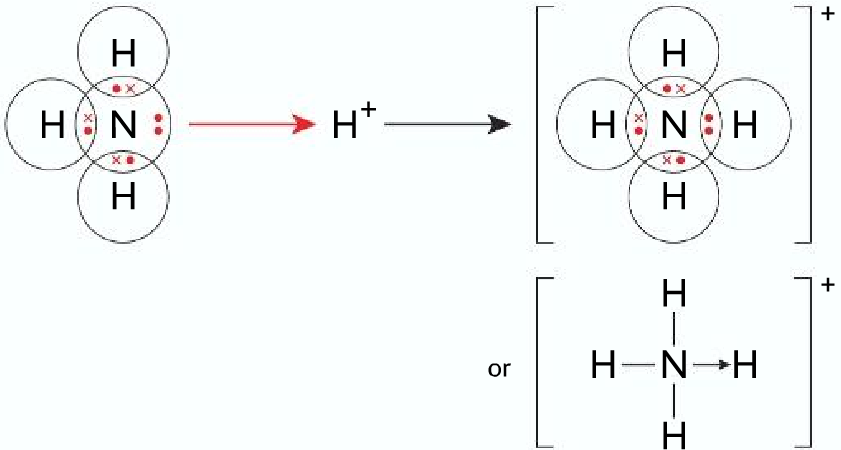
In a normal covalent bond, each electron in the shared electron pair comes from different atoms. But in a dative covalent bond the shared electron pair comes from the same atom. Both electrons in the shared pair are from the same atom. For example, ammonia forming a dative covalent bond with a hydrogen ion.
In ammonia, the nitrogen atom has a lone pair electron (unused electron pair), which it donates to the H+ ion to form a dative covalent bond. The dative covalent bond is represented by a ⟶ instead of a – . Compound that has lone pair electrons can form a dative covalent bond.
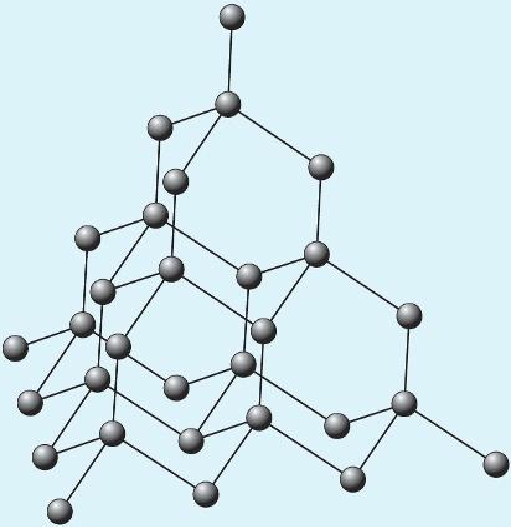
Giant Covalent Structures
In giant covalent structure, the covalent bonds continue throughout the whole structure. Examples of giant covalent structure: diamond, silicon (IV) carbide, and silicon (IV) oxide SiO2 . Giant covalent structures have high melting temperatures because strong covalent bonds continue throughout the whole structure, which takes a very high energy to break
Shapes of covalent molecules
When electron pairs exist around the central atom in a covalent compound they repel each other. The electron pairs around the central atom take up a shape which minimizes the repulsion between them. Different covalent compounds take up different shapes depending on the number of lone pairs around them.
Examples:
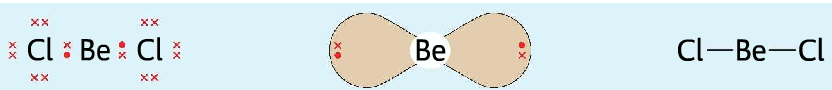
- In BeCl2 molecule, the central atom has two electron pair around it. It takes up a linear shape.
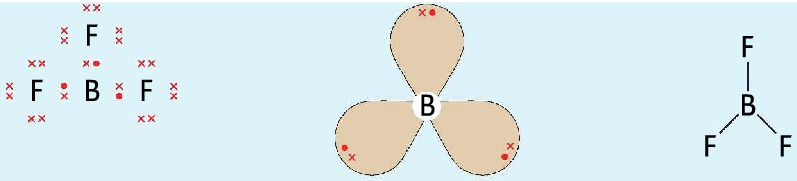
- In BF3 molecule, the central atom has three pairs of electrons around it. Takes up a trigonal planar shape
- In H2O molecules, there are four electron pairs around the O atom, so takes up a bent shape.
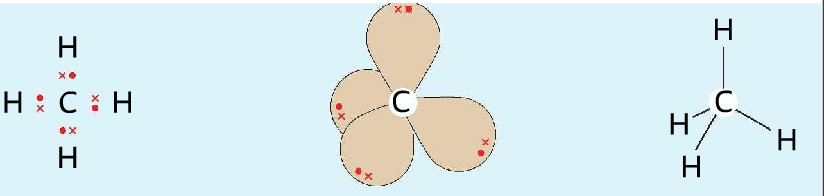
- In methane, there are four electron pairs around the central carbon atom. Takes up the tetrahedral shape


Deference to op, some wonderful entropy.