The concept of mole is a very important part of chemistry. Mole makes calculations in chemistry so much easier! But the different concepts of mole are even more important when you move on from IGCSE chemistry to IAL Chemistry. Unlike IGCSE chemistry the mole is not confined to only a chapter. In IAL chemistry you will need to use the concepts of mole in almost ALL of the chapters to calculate different things such as concentration, mass of product produced, volume of gas produced, in enthalpy changes and so much more!
Even though it is extremely important to understand the mole and have a crystal clear understanding about it, most of the text books does very little to simplify things to the students. All they give are generic definitions, formulas, terms etc. which confuses the students even more. No real life examples with which students can relate to and understand.
Even when I as a student had a really tough time understanding mole, my teacher only provided me with printed notes to memorize. It is only when I started to teach chemistry I dissected the different concepts of moles and had a crystal clear understanding.
In this blog post my main goal would be to present to you the different concepts/basics of mole in simple words. We are going to start right from scratch. We are going to understand what each definition mean, what does Avagadro’s number mean, why we use mole in chemistry, different examples of how mole is used, math and so much more!
What does “Mole” mean in Chemistry
When you hear the word “Mole” you might think about a small hairy animal or just a big dark spot on your body. But in chemistry the mole is a completely different thing. The biggest mistakes text books do is trying to explain moles using definitions. Most students hardly understand definitions, they memorize them and the different formulas which follows. Let me explain you mole the simplest way possible! So here we go! The mole is nothing but a huge number and that huge number is 6.02 x 1023, this huge number is also called Avagadro’s number!

1 Mole = 6.02 x 1023
In chemistry, “mole” is simply a HUGE number! 1 mole simply equals 6.02 x 1023
This huge number is also called Avagadro’s number or Avagadro’s Constant. That’s it!
A real life example to understand mole better

What can be a better example than eggs? What do we call 12 eggs? 1 dozen right? 1 dozen = 12. The term dozen represents a number, which is 12. Similarly the word “mole” also represents a number, but much bigger, which is 6.02 x 1023.
- 1 dozen of eggs = 12 eggs
- 1/2 dozen of eggs = 12/2 = 6 eggs
- 1 mole of eggs = 6.02 x 1023 eggs
- 1/2 mole of eggs = 6.02 x 1023/2 = 3.01 x 1023 eggs
You can see that the concept of mole is very similar to how we use the concept of dozen when buying eggs
Why use mole in Chemistry?
Now you might wonder what’s the point of using the concept of mole? In chemistry we have to keep track of the number of particles, but there are a lot! Even in a single grain of salt or sugar there are billions and billions of particles. So we need a way to keep track of the particles, like how many reactant particles reacted with each other, how many reactant particles produced etc.
But if we normally try to keep track of the particles it becomes quite difficult. Let me give you a simple example, for example there are 30 students in a class in school, we can easily represent 150 by just saying students from 5 classes will come to the party. The concept of mole also acts an intermediate volume, mass, no. of particles etc. But don’t worry we will have a look at those things later on this blog post. In the meantime, lets have a look how more makes things easier.
- 1.204× 1024 sodium atoms means 2 mol sodium
- 3.01× 1023 molecules of water means 0.5 mol of water
- 5 mole of calcium means 3.01 x 10 24 atoms of calcium
- 3 mole of oxygen means 1.806 x 10 24 molecules of oxygen
You can already see from the simple examples above that how using mole makes things so much shorter and easy to understand. Of course I will also show you step by step of how you can find out the number of moles and get the number of particles from mole in this blog, so please do not worry!
To learn more about the concepts of moles you can check out this awesome book “You Can Do Chemistry: Moles & Stoichiometry” on Amazon.
Calculating Number of moles from number of particles
We will repeat this “how to calculate moles” quite a lot in this blog. This will clear up your concepts if we do it several times instead of all at once. So that now we are clear that why we use moles in chemistry, let’s go on and learn how we can find the number of moles from the number of particles or calculate the number of particles from the number of moles.
Examples:
1) Find out the number of moles for 1.204 x 1024 atoms of sodium atoms
6.02 x 1023 atoms of sodium means 1 mole of sodium
Then 1.204 x 1024 sodium atoms in (1.204 x 1024)/6.02 x 1023 = 2 mole of sodium
2) Find out the number of particles in 5 mole of calcium
1 mole of calcium means 6.02 x 1023 atoms calcium
So in 5 mole of calcium the number of atoms are = 5 x (6.02 x 1023) = 3.01 x 1024
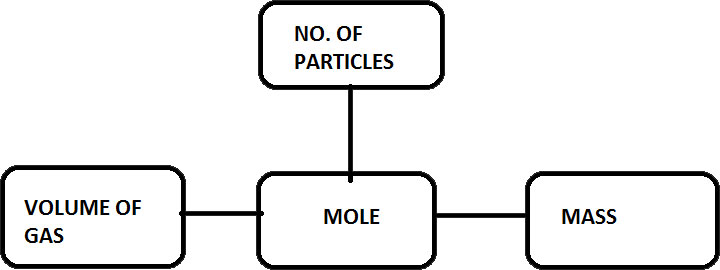
Mole connects mass, volume of gas and number of particles
Mole plays a vital role in linking the mass of a substance, volume of gas and the number of particles present. There are a certain pieces or sets of rules which easily lets us to find out the mass of a certain volume of gas, or to find out the number of particles in a certain mass of substance. All thanks to the concepts of moles. You are going to have a look at it step by step of how it is done.
Here is the definition of mole:
“ A mole of any substance is defined as the amount of substance that contains as many particles (atoms, ions or molecules as there are atoms in exactly 12 g of carbon-12”
Breakdown of the definition of mole:
- 1 mole carbon-12 contains 6× 1023 atoms of carbon, and weighs exactly 12g
- The mass of each carbon atom is 12 atomic units.
- Suppose if we have 1 mole of atoms each of mass 1 atomic unit
- Then the mass of 1 mole of those atoms will be 1 g
Connecting Mole with mass
The mass of one mole of any substance is called the “Molar Mass”
The mass of one mole of any element or compound is it’s RAM (Relative Atomic Mass) or RFM (Relative Formula Mass) with gram.
For example: The Relative Atomic Mass of sodium is 23, so if we take one mole of sodium it’s mass would be 23g. You can find out the relative atomic mass from the periodic table. If your concepts with periodic table are weak then you can have a quick look at my periodic table blog. So the Molar mass of sodium is 23g.
Molar mass also have a symbol so we can write is quickly, it is M .
Hence we can write M (Na) = 23g
Another Example: suppose the relative atomic mass of Magnesium is 24, hence the mass of one mole of magnesium is 24g. In another word we can say the Molar Mass of Magnesium is 24g.
M (Mg) = 24g
Now let’s move on to Molar Mass of compounds.
The Molar Mass of compounds is their relative formula mass/relative molecular mass ( sum of all the relative atomic masses in the compound)
For Example: the formula of sodium chloride is NaCl, sodium has a Relative Atomic Mass of 23 and Chlorine has a relative atomic mass of 35.5.
So their relative formula mass will be = 23 + 35.5 = 58.5
Hence the mass of 1 mole of sodium chloride will be = 58.5 g
The molar mass of Sodium Chloride is 58.5g
M (NaCl) = 58.5 g
Examples:
1) 0.5 mole of Magnesium weighs?
1 mole Mg —- 24g
0.5 mole Mg — 24× 0.5 = 12g
2) 0.25g of CO2 is how many mole?
1 mole of CO2 weighs — (12+16× 2) = 44𝑔
44g CO2 ———— 1 mole CO2
1 g CO2 ———- 1/44 mole CO2
0.25g of CO2 ———- (1/44) × 0.25 = 0.0057 mole
Calculating Number of particles from mass using mole concept
The concept of mole makes it extremely easy for us to calculate the number of particles present in a given mass of substance. This concept will be absolutely cleared up once you have a look at the following example below.
Example: Find the number of particles in 3g of calcium
Upto now, now we know how to find the mass of one mole of a substance. We can easily find out the number of particles in 3g of calcium without using any type of formula, just simple concepts.
The Relative Atomic Mass of Calcium is 40, hence the molar mass of calcium is 40g, we can directly write 1 mole of calcium as 40g
1 Mole of Calcium consists of 6.02 × 1023 atoms
We can also write 1 mole as 40g, so
40g of Calcium consists of 6.02 × 1023 atoms
1g of calcium consists of (6.02 × 1023)/40 atoms
Hence,
3g of calcium consists of (6.02 × 1023)/40 x 3 = 4.52 × 1022 atoms of calcium
Now let’s have a look at another example, it is opposite of the previous one. In this example it is required to find out the mass of a certain number of particles. This concept will be cleared up once you have a look at the simple example below.
Example: Find the mass of 4× 1012 atoms of potassium
The molar mass of calcium is 39.1g (by now you should know how I found it)
Mass of 1 mole of Potassium is 39.1 g
6× 1023 atoms of potassium weighs 39.1 g ( 1 mole = 6× 1023 particles)
1 atom potassium weighs (39.1/6× 1023)g
4× 1012 atoms of potassium weighs (39.1/6× 1023) x 4× 1012 = 2.6× 10-10 g
Avagadro’s Gas Law- Connecting mole with volume of gas
Avagadro must have been a very cool person, he has a big number and a law named after him! But even though the name of the law may sound fancy and difficult to understand but the concept is really easy. This law will help you to find out the number of moles of a certain volume of gas, also their mass! But of course there are certain assumptions which you will have to keep in mind. So let’s have a look at the bookish version of the definition of “Avagadro’s Law” and then my interpretation.
Here is the bookish version of Avagadro’s Law:
“ Equal volumes of all gases contain equal numbers of molecules, provided that they are at the same temperature and pressure”
Breakdown of Avagadro’s Law
- At the same same temperature and pressure if two gases have same volume, then they will also have the same number of particles.
- JUST AN EXAMPLE: suppose, both gases carbon dioxide and oxygen gas have a volume of 20 cm3 at 20 degree celcius and 1 atm pressure. Suppose carbon dioxide has 150 gas particles, then according to Avagardo’s Gas law, oxygen will also have 150 gas particles.
- ANOTHER EXAMPLE: suppose two gases are at the same temperature and pressue, but the volume of one gas is double the volume of the other gas, then that gas will also have double the numer of particles.
- Suppose, both sulfur dioxide and fluorine gas are at the same temperature and pressure (20 degree Celsius and 1 atm pressure), but the volume of sulfur dioxide is 40 cm3 while the volume of fluorine is 20 cm3. So if fluorine has 150 gas particles, then according to the gas law sulfur dioxide will have double the number of particles, which is 300 particles.
What is “Molar Volume”?
Molar volume is a term obtained from the Avagadro’s gas law. It is a very simple but useful concept. The concept is very similar to the molar mass. The molar volume is the volume of one mole of gas at 1 atm pressure and 298 K temperature. The value of molar volume is 24 dm3 or 24,000 cm3. ANY 1 mole of gas at 1 atm pressure and 298K temperature will have a volume of 24dm3 or 24,000 cm3.
For example, 1 mole of carbon dioxide gas and 1 mole of oxygen gas at 1 atm pressure and 298K will both have a volume of 24 dm3 or 24,000 cm3.
Example: Finding volume of gas from a given mass
Find the volume of 2g of CO2(Carbon dioxide) gas
Volume of 1 mole of CO2 is 24,000 cm3
Volume of 44g of CO2 is 24,000 cm3
Volume of 1g of CO2 is (24,000 cm3 / 44)
Hence the volume of 2g of CO2 is (24,000 cm3/44) x 2 = 1090cm3
Example: Converting Volume of Gas to Mass using mole concepts
Find the mass of 150 cm3 hydrogen gas
1 mole of hydrogen weighs 2g (note: hydrogen exists as a diatomic molecule H2)
24,000 cm3 of hydrogen gas will weigh 2g
1 cm3 of hydrogen will weigh (2/24,000)
Hence 150 cm3 of hydrogen will weigh (2/24,000) x 150 = 0.0125g
Practice!
1. Calculate the number of moles in:
8g of Oxygen-16 atoms
48g of Carbon-12 atoms
8.88g of Chlorine atoms
2. Calculate the number of atoms in:
36 g of carbon-12
4g of magnesium-24
532g of caesium-133
57g of lead-208
Conclusion
Thank you for reading this blog post. In this blog post we have covered all the basic concepts of mole from scratch. I hope you have got a lot of value out of this. The mole is a very important topic in IAL Chemistry so I will definitely write more blog posts on it. Until then study smart and not hard!


For the following example, the answer you have given is incorrect.
Example: Find the number of particles in 3g of calcium
the correct answer is 4.52 x 10^22 and not 4.52 x 10^23
Thank you for your feedback, I have corrected the answer.